
Used by the NHS
to treat depression
Flow is now active across 6 NHS trusts. Each has independently published results showing it works, safely and consistently.
A Crisis Service reported around 80% of patients reported a decrease in their depressive symptoms1.
"I’ve seen people who haven’t responded to anything else, no longer struggle with depression.”
Dr Mark McConnochie
NHS Consultant Psychiatrist
Proven and safe
Backed by 25+ years
of research.
Flow delivers gentle electrical stimulation to the part of your brain that regulates mood. This technology, called tDCS, has been used in clinical settings for over 25 years and proven safe and effective across multiple independent trials.
Users feel better in 3 weeks2
77%
Average time to notice changes3
3 weeks
Depression-free by week 104
57 %
Flow
Is it right for you?
Take a 2-minute quiz to find out if Flow could help with your symptoms of depression.
Take the quiz

Order your Flow headset
Accessible in one click, delivered to your door within 1-3 working days.
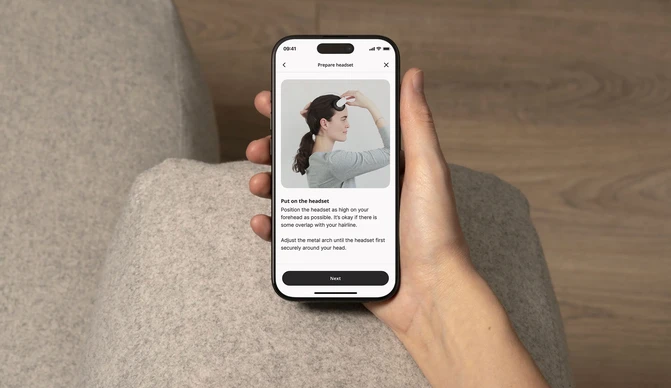
Download the app & start your plan
Get guided daily therapy and brain stimulation sessions, backed by clinical science.

Track your progress & feel the shift
Start noticing results as early as week 3, with full support from our team.
Flow in Action
UK
Global overview
See clinical improvements within 3 weeks
77 %
Real people. Real results.
Stories from people living with depression, and what changed.
57% of Flow users no longer have depression at 10 weeks3. These are their stories.
“The world was in colour again”
Annie,
25, Aylesbury, United Kingdom
"Flow's been the ladder out of a very dark hole"
Jon,
35, London, United Kingdom
In the press
Do you have any questions?
Footnote
[1] Leicestershire Partnership NHS Trust (LPT), Mar 2023
[2] Flow real-world data (n=14,383, Apr 2025)
[3] Flow real-world data (n=14,383, Apr 2025)
[4] Real world data, an improvement of 3pts or more on the clinically validated MADRS-S survey after 3 weeks, n=14,383. Woodham, R.D., Selvaraj, S., Lajmi, N. et al. Nat Med (2024).
The Flow headset is proven and safe when used as instructed








